CALLING ALL CHEMISTRY MAJORS/GURU
I'll take a shot...its been over a decade but I am pretty sure it does make a difference because if you were to translate back you are dealing with different bonds, which would change the compound (like ethane vs methane etc.) I think. Consult your chem book. I have my notes in the attic.
Originally Posted by C U AT 9K,Sep 29 2007, 07:21 PM
I'd help you but I can't understand that molecule. You have H coming off of H??
It looks like this:
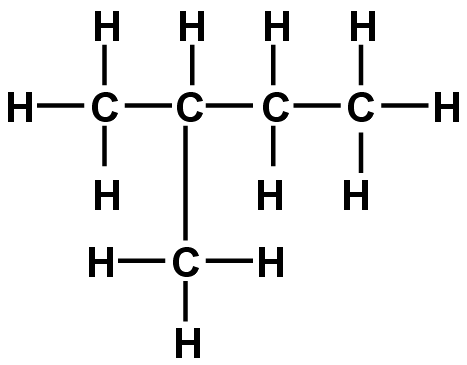
Methylbutane I believe.
CH3CH(CH3)CH2CH3
You are supposed to write the carbons in sequence. Start by finding the longest chain of carbons (in this case, 4). Begin at the end closest to the first functional group. That extra CH3 (the methyl group) is off the SECOND carbon which means you must first write CH3 to account for the first one. Then its CH(CH3), and so on. You'll get it wrong if you don't write it in order.
2-methyl butane
You are supposed to write the carbons in sequence. Start by finding the longest chain of carbons (in this case, 4). Begin at the end closest to the first functional group. That extra CH3 (the methyl group) is off the SECOND carbon which means you must first write CH3 to account for the first one. Then its CH(CH3), and so on. You'll get it wrong if you don't write it in order.
2-methyl butane
Trending Topics
Originally Posted by Seks,Sep 29 2007, 12:20 PM
H H H H
| | | |
H - C - C - C - C - H
| | | |
H | H H
|
H - C - H
|
H
Any diff if I write the above as (CH3)2CHCH2CH3 or CH(CH3)2CH2CH3?????
| | | |
H - C - C - C - C - H
| | | |
H | H H
|
H - C - H
|
H
Any diff if I write the above as (CH3)2CHCH2CH3 or CH(CH3)2CH2CH3?????
The second one is wrong as well because you have 3 hydrogens coming off the first carbon, not one. Also, the methyl group is coming off the SECOND carbon.
Originally Posted by magician,Sep 30 2007, 03:34 PM
Isn't the "2-" superfluous?




