put heat wrap on my intake
#1
Registered User

Thread Starter
Join Date: Feb 2014
Location: oakville
Posts: 214
Likes: 0
Received 0 Likes
on
0 Posts
#3
Registered User

Thread Starter
Join Date: Feb 2014
Location: oakville
Posts: 214
Likes: 0
Received 0 Likes
on
0 Posts
It's a barrier between two temperatures, the hotter one stays on one side of the barrier, and the other side is unaffected in a perfect world.
In this case, the intake/intake tube temperature is one side of the barrier, and the under-hood temp is the other side of the barrier. Like wearing an oven mitt and putting your hand in the oven. you won't feel the heat from the oven on your hand.
.
the issue is that when the barrier itself heats up it turns into a heat blanket wrapping the intake tube, and will take much longer to lose temp (making the heat soaking worse). now the question is how long would it take until it would get to 100% of the under-hood temp? Or would it be like an oven mitt where you could stand there holding a 300 degree plate for as long as you want to?
#5
Registered User

Thread Starter
Join Date: Feb 2014
Location: oakville
Posts: 214
Likes: 0
Received 0 Likes
on
0 Posts
http://i279.photobucket.com/albums/k...psdbdacde4.jpg
#7

Originally Posted by starchland' timestamp='1425350585' post='23525202
Look at f1 intakes. They use a wrap designed to reflect radiant heat
http://i279.photobucket.com/albums/k...psdbdacde4.jpg
Trending Topics
#9
Registered User

Plastic is a good insulator, if maybe it was metal it might help.
If you're doing it for looks then just do it, what does it matter.
I think it looks good with just the stock arm.
If you're doing it for looks then just do it, what does it matter.
I think it looks good with just the stock arm.
#10
As as designer of gas turbine intakes and exhausts, I would hesitate to wrap the intake with insulation, unless the you could guarantee a constant flow of cold, dense air into the engine. Once the engine is switched off, the engine bay remains hot. Eventually, the intake system will heat soak and be at the same ambient temperature as the rest of the engine bay. So now, the insulation you have put around the intake is keeping the heat in and warming up your intake air, reducing engine output, by decreasing the intake air density. You could calculate this with the following:
The air mass in is directly related to the density of air, and the introduced volume.
 ,where
,where 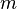 is the mass,
is the mass, 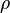 is the density and
is the density and  is the volume of the gas. As the volume
is the volume of the gas. As the volume  is fixed, only density
is fixed, only density 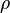 of the air can be modified to vary air mass. The density of the air depends of the relative humidity, altitude, pressure drop and temperature.
of the air can be modified to vary air mass. The density of the air depends of the relative humidity, altitude, pressure drop and temperature.
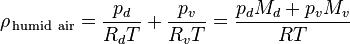 where:
where:
 Density of the humid air (kg/m³)
Density of the humid air (kg/m³) Partial pressure of dry air (Pa)
Partial pressure of dry air (Pa) Specific gas constant for dry air, 287.058 J/(kg·K)
Specific gas constant for dry air, 287.058 J/(kg·K) Temperature (K)
Temperature (K) Pressure of water vapor (Pa)
Pressure of water vapor (Pa) Specific gas constant for water vapor, 461.495 J/(kg·K)
Specific gas constant for water vapor, 461.495 J/(kg·K) Molar mass of dry air, 0.028964 (kg/mol)
Molar mass of dry air, 0.028964 (kg/mol) Molar mass of water vapor, 0.018016 (kg/mol)
Molar mass of water vapor, 0.018016 (kg/mol) Universal gas constant, 8.314 J/(K·mol)
Universal gas constant, 8.314 J/(K·mol)
If you are worried, you would be better off putting a heat shield between the engine and the intake.
The air mass in is directly related to the density of air, and the introduced volume.
 ,where
,where 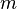 is the mass,
is the mass, 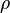 is the density and
is the density and  is the volume of the gas. As the volume
is the volume of the gas. As the volume  is fixed, only density
is fixed, only density 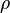 of the air can be modified to vary air mass. The density of the air depends of the relative humidity, altitude, pressure drop and temperature.
of the air can be modified to vary air mass. The density of the air depends of the relative humidity, altitude, pressure drop and temperature.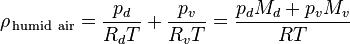 where:
where: Density of the humid air (kg/m³)
Density of the humid air (kg/m³) Partial pressure of dry air (Pa)
Partial pressure of dry air (Pa) Specific gas constant for dry air, 287.058 J/(kg·K)
Specific gas constant for dry air, 287.058 J/(kg·K) Temperature (K)
Temperature (K) Pressure of water vapor (Pa)
Pressure of water vapor (Pa) Specific gas constant for water vapor, 461.495 J/(kg·K)
Specific gas constant for water vapor, 461.495 J/(kg·K) Molar mass of dry air, 0.028964 (kg/mol)
Molar mass of dry air, 0.028964 (kg/mol) Molar mass of water vapor, 0.018016 (kg/mol)
Molar mass of water vapor, 0.018016 (kg/mol) Universal gas constant, 8.314 J/(K·mol)
Universal gas constant, 8.314 J/(K·mol)If you are worried, you would be better off putting a heat shield between the engine and the intake.






